
Chemistry, 10.10.2019 19:30, LikeIke2859
According to the equation 2na + 2h2o mc012-1.jpg 2naoh+h2, what mass of na is required to yield 22.4 l of h2 at stp? (the atomic mass of na is 22.99 u.)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, 20alondra04
In order to calculate the amount of heat transferred you must know the __ and specific heat of the material, as well as the change in temperature. a. volume b. density c. mass d. enthalpy
Answers: 1


Chemistry, 22.06.2019 17:10, sophiaa23
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1

Chemistry, 23.06.2019 11:00, randyg0531
Which of the following reactions is endothermic? h2(g) + ½ o2(g) h2o(g), h = -57.82 kcal ½n2(g) + o2(g) + 8.1 kcal no2(g) ½ n2(g) + 3/2 h2(g) nh3(g) + 11.0 kcal c(diamond) + o2(g) co2, h = -94.50 kcal
Answers: 2
Do you know the correct answer?
According to the equation 2na + 2h2o mc012-1.jpg 2naoh+h2, what mass of na is required to yield 22.4...
Questions in other subjects:

Mathematics, 25.08.2020 21:01




Social Studies, 25.08.2020 21:01

Mathematics, 25.08.2020 21:01

Mathematics, 25.08.2020 21:01


Mathematics, 25.08.2020 21:01



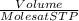
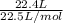
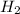 : Na is 1 : 2
: Na is 1 : 2



