Which of the following best describes what happens to the substance between t4 and t5?
...

Which of the following best describes what happens to the substance between t4 and t5?
(a) the molecules are leaving the liquid phase.
(b) the solid and liquid phases coexist in the equilibrium.
(c) the vapor pressure of the substance is decreasing.
(d) the average intermolecular distance is decreasing.
(e) the temperature of the substance is increasing.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, itzhari101
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 10:40, yfgkeyonna
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 10:50, lejeanjamespete1
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 11:00, justarando
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3
Do you know the correct answer?
Questions in other subjects:


Biology, 01.02.2021 18:30



Mathematics, 01.02.2021 18:30

Mathematics, 01.02.2021 18:30




Mathematics, 01.02.2021 18:30


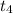 and
and 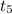 , the molecules are leaving the liquid phase.
, the molecules are leaving the liquid phase. 




