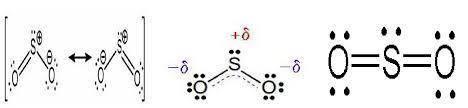
The compound that can best be represented using two or more resonance forms is SO2.
Sometimes, a single Lewis structure is insufficient to account for certain observed properties of molecules such a bond length that is intermediate between the values of a single and double bond and fractional bond orders.
In such cases, we must assume that the actual structure of the molecule lies in-between hypothetical bonding extremes called resonance structures or canonical structures.
Giving the options in the questions, canonical structures only arise for SO2 molecule as shown in the image attached to this answer.
Learn more: link