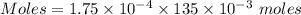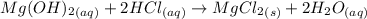Chemistry, 14.01.2020 19:31, hnsanders00
When 220 ml of 1.50x10^-4 m hydrochloric acid is added to 135 ml of 1.75x10^-4 m mg(oh)2, the resulting solution will

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, shafferakr6
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 22.06.2019 01:00, chrisxxxrv24
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 22.06.2019 08:30, Blaise2653
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1
Do you know the correct answer?
When 220 ml of 1.50x10^-4 m hydrochloric acid is added to 135 ml of 1.75x10^-4 m mg(oh)2, the result...
Questions in other subjects:


Chemistry, 05.05.2020 06:06








Mathematics, 05.05.2020 06:06



 M
M
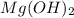 :
:
 M
M