

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:20, maevemboucher78
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.
Answers: 2

Chemistry, 22.06.2019 00:30, S4NCHEZ28
Sarah wants to know where in her garden chamomile would grow the best. she thinks chamomile will grow best in the corner of the garden that gets the most sunlight. to test her hypothesis, she decides to plant several groups of chamomile in her garden as an experiment. which of the following variables will sarah need to measure to know which group of plants grew best? a. the location of the plants b. the type of plants c. the height of the plants d. the amount of water she gives the plants
Answers: 1

Chemistry, 22.06.2019 11:00, coco8560
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 16:00, esme06quirino
Is a measure of the resistance to flow. a high liquid has a high resistance to flow and flows slowly. the ancients thought everything in the world was made of 4 we now know that there are 94 naturally occurring and scientists have created another 24 i am certain they will create even more. honey flows slowly because it has a high to flowing. a can be separated by physical means because it contains more than one pure substance and 2 pure substances are not chemically bonded to each other. a cannot be separated by physical means. all matter is made up of all elements are with the same number of protons. if it is just a single or many bonded together, if all of them have the same number of protons, it is an element. in a piece of pure iron metal, all the are joined together, that piece of iron metal is called elemental iron. a single of iron is called elemental iron. a mixture has differences from place to place. we might need a microscope to see them or they might be obvious to the unaided eye. there are surfaces separating it into different phases. a mixture is the same everywhere. it is uniform. there are no surfaces separating it into different phases. if different kinds of atoms (different elements) are bonded together by their electrons, it is called a there are physical means of to isolate the different pure substances in a mixture and there are chemical means of to isolate the different elements in a compound. 1. element 2. compound 3. mixture 4. heterogeneous 5. homogeneous 6. pure substance 7. atoms 8. separation 9. viscosity 10. resistance
Answers: 2
Do you know the correct answer?
Consider the reaction 2 no2(g) + f2(g) => 2 no2f(g) rate = -δ[f2]/δtwhat is another form of the...
Questions in other subjects:











![rate=-\frac{ d[F_{2}]}{dt}=-\frac{1}{2}*\frac{ d[NO_{2}]}{dt}=\frac{1}{2}*\frac{d[NO_{2}F]}{dt}](/tpl/images/0453/2658/87f2e.png)
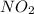 react with one molecule of
react with one molecule of 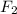 to produce two molecules of
to produce two molecules of 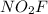 .
. ![rate=-\frac{ d[F_{2}]}{dt}](/tpl/images/0453/2658/f5227.png) , in other words as the consumption of
, in other words as the consumption of ![rate=-\frac{1}{2}*\frac{ d[NO_{2}]}{dt}](/tpl/images/0453/2658/62960.png) , with negative sign for consumption and the (1/2) to normalize the rate. Note that as the stoichiometric coefficient it's two, the rate of
, with negative sign for consumption and the (1/2) to normalize the rate. Note that as the stoichiometric coefficient it's two, the rate of ![rate=\frac{1}{2}*\frac{d[NO_{2}F]}{dt}](/tpl/images/0453/2658/ad022.png) .
.



