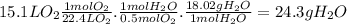What mass of water, in grams, would have to be decomposed
toproduce 15.1 l of molecular oxygen...

Chemistry, 09.01.2020 07:31, heyperdomo8496
What mass of water, in grams, would have to be decomposed
toproduce 15.1 l of molecular oxygen at stp?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:00, yaneiryx5476
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1


Chemistry, 22.06.2019 18:00, jeepjose58
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Mathematics, 26.11.2020 22:10



History, 26.11.2020 22:10

History, 26.11.2020 22:10

Physics, 26.11.2020 22:10

French, 26.11.2020 22:10

Biology, 26.11.2020 22:10