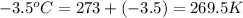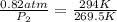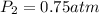Chemistry, 30.08.2019 12:30, brandiwingard
Atire at 21°c has a pressure of 0.82 atm. its temperature decreases to –3.5°c. if there is no volume change in the tire, what is the pressure after the temperature change?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 19:30, 2020sanchezyiczela
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3
Do you know the correct answer?
Atire at 21°c has a pressure of 0.82 atm. its temperature decreases to –3.5°c. if there is no volume...
Questions in other subjects:


English, 11.12.2020 04:30







Mathematics, 11.12.2020 04:30

Mathematics, 11.12.2020 04:30


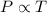 (At constant volume and number of moles)
(At constant volume and number of moles)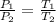
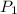 = initial pressure = 0.82 atm
= initial pressure = 0.82 atm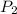 = final pressure = ?
= final pressure = ?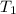 = initial temperature =
= initial temperature = 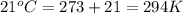
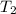 = final temperature =
= final temperature =