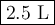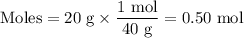Chemistry, 04.01.2020 01:31, MsShreve9939
The molarity of a sodium hydroxide (naoh) solution is 0.2 m. the molar mass of naoh is 40 g/mol. if the solution contains 20 g of sodium hydroxide dissolved in water, what is the volume of the solution?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, ksalinas7404
Un cierto gas tiene un volumen de 800ml a 80°c y 600ml a 80°c y 600mmhg de presión. ¿cual será el volumen del gas a condiciones normales? sí el gas es oxígeno, ¿cuál será su peso? y ¿cuántas moléculas están presentes en el sistema?
Answers: 2



Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2
Do you know the correct answer?
The molarity of a sodium hydroxide (naoh) solution is 0.2 m. the molar mass of naoh is 40 g/mol. if...
Questions in other subjects:

Mathematics, 24.03.2021 23:20

English, 24.03.2021 23:20

Mathematics, 24.03.2021 23:20

Mathematics, 24.03.2021 23:20

Mathematics, 24.03.2021 23:20

Mathematics, 24.03.2021 23:20

Mathematics, 24.03.2021 23:20


Social Studies, 24.03.2021 23:20