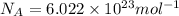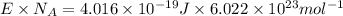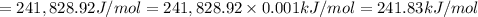Chemistry, 03.01.2020 20:31, abraralzaher
The longest wavelength of light with enough energy to break the cl-cl bond in cl2(g) is 495 nm.
1) calculate the frequency in s-1 of the light2) calculate the energy in j of a photon of the light3) calculate the minimun energy in kj mol-1 of the cl-cl bond

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:00, luffybunny
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1

Chemistry, 22.06.2019 18:30, bibiansolis
The table lists the lattice energies of some compounds. compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf. the lattice energy increases as the cations get larger, as shown by lif and licl. the lattice energy decreases as cations get smaller, as shown by nacl and naf. the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 23.06.2019 00:00, maronetham6253
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2

Chemistry, 23.06.2019 01:00, birdman2540
Which of the following is in the lanthanide family? a) uranium b) promethium c) silver d) gold
Answers: 2
Do you know the correct answer?
The longest wavelength of light with enough energy to break the cl-cl bond in cl2(g) is 495 nm.
Questions in other subjects:

Mathematics, 19.04.2021 20:50

Mathematics, 19.04.2021 20:50

Biology, 19.04.2021 20:50




Computers and Technology, 19.04.2021 20:50



Computers and Technology, 19.04.2021 20:50


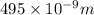
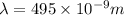
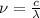
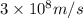
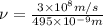
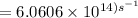
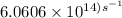 .
.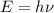
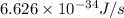
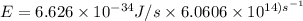

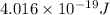 .
.