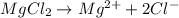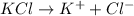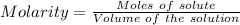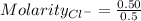Chemistry, 02.01.2020 23:31, eggoysters
A0.20 mol sample of mgcl2(s) and a 0.10 mol sample of kcl(s) are dissolved in water and diluted to 500 ml. what is the concentration of cl- in the solution?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, micvar9646
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 04:50, aletadaboss
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 06:30, luhmimi17
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 23.06.2019 01:30, Elliendc7939
List and describe the neurological effects of the vocs and other air pollutants, as described by dr. theo colborn
Answers: 2
Do you know the correct answer?
A0.20 mol sample of mgcl2(s) and a 0.10 mol sample of kcl(s) are dissolved in water and diluted to 5...
Questions in other subjects:

Spanish, 25.12.2019 11:31



Mathematics, 25.12.2019 11:31


Mathematics, 25.12.2019 11:31



History, 25.12.2019 11:31