
Chemistry, 28.12.2019 03:31, camila9022
Determine the heat of reaction (δhrxn) for the combustion of ethanol (c2h5oh) by using heat of formation data: c2h5oh (l) + 3 o2 (g) → 2 co2 + 3 h2o (g)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, BigDough9090
Becquerel expected to find ( he developed the photographic plate that had sun-exposed minerals on top of it. becquerel expected to find ( he developed the photographic plate that had been in the closed drawer.
Answers: 2

Chemistry, 22.06.2019 03:30, ilizzy1224
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3


Chemistry, 22.06.2019 20:40, ohgeezy
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
Do you know the correct answer?
Determine the heat of reaction (δhrxn) for the combustion of ethanol (c2h5oh) by using heat of forma...
Questions in other subjects:

Mathematics, 02.10.2020 15:01


Computers and Technology, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01

Chemistry, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01

Business, 02.10.2020 15:01

Chemistry, 02.10.2020 15:01

Biology, 02.10.2020 15:01


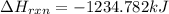
![\Delta H_{rxn}=\sum [n_{i}\times \Delta H_{f}^{0}(product)_{i}]-\sum [n_{j}\times \Delta H_{f}^{0}(reactant_{j})]](/tpl/images/0435/2901/43912.png)
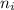 and
and 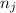 are number of moles of product and reactant respectively (equal to their stoichiometric coefficient).
are number of moles of product and reactant respectively (equal to their stoichiometric coefficient).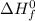 is standard heat of formation.
is standard heat of formation.![\Delta H_{rxn}=[2mol\times \Delta H_{f}^{0}(CO_{2})_{g}]+[3mol\times \Delta H_{f}^{0}(H_{2}O)_{g}]-[1mol\times \Delta H_{f}^{0}(C_{2}H_{5}OH)_{l}]-[3mol\times \Delta H_{f}^{0}(O_{2})_{g}]](/tpl/images/0435/2901/adad7.png)
![\Delta H_{rxn}=[2mol\times -393.509kJ/mol]+[3mol\times -241.818kJ/mol]-[1mol\times -277.69kJ/mol]-[3mol\times 0kJ/mol]](/tpl/images/0435/2901/6491b.png)





