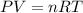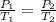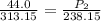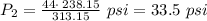Chemistry, 28.12.2019 03:31, brooke0713
The pressure inside an automobile tire is 44.0 pounds per square inch (psi) on a very warm summer day (40.0 °c). calculate the tire pressure in the same tire on a very cold winter day (-35.0 °c) five months later, assuming that no air escapes from the tire. hint: you will need to make another reasonable assumption that is not stated here.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 23:00, lufung8627
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1


Chemistry, 23.06.2019 07:00, jstyopin
In order for a high temperature boiler or steam engine to produce superheated water, or steam: the heat source must be greater than 100°c the water must be permitted to evaporate quickly the system must be sealed and become pressurized above atmospheric pressure the vapor pressure must be kept below 760 mm(hg)
Answers: 1

Chemistry, 23.06.2019 13:30, sophie5988
Where are electrons with the lowest energy found? in the nucleus farthest from the nucleus outside the atom closest to the nucleus
Answers: 1
Do you know the correct answer?
The pressure inside an automobile tire is 44.0 pounds per square inch (psi) on a very warm summer da...
Questions in other subjects:

Mathematics, 18.12.2020 21:40

Mathematics, 18.12.2020 21:40

Law, 18.12.2020 21:40

Mathematics, 18.12.2020 21:40



Mathematics, 18.12.2020 21:40