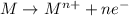Atoms of elements at the top of a group on the periodic table are smaller than the atoms of elements at the bottom of the group. how does this explain the difference in the reactivity of metals within a group?
the valence electrons of the larger atoms are farther from the nucleus and are easier to remove, so the metals near the top are more reactive than those at the bottom.
the valence electrons of the larger atoms are farther from the nucleus and are harder to remove, so the metals near the bottom are more reactive than those at the top.
the valence electrons of the larger atoms are farther from the nucleus and are harder to remove, so the metals near the top are more reactive than those at the bottom.
the valence electrons of the larger atoms are farther from the nucleus and are easier to remove, so the metals near the bottom are more reactive than those at the top.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:50, mrylenastewart
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 07:00, shradhwaip2426
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3
Do you know the correct answer?
Atoms of elements at the top of a group on the periodic table are smaller than the atoms of elements...
Questions in other subjects:

Mathematics, 08.05.2021 02:50


Mathematics, 08.05.2021 02:50

Mathematics, 08.05.2021 02:50

Mathematics, 08.05.2021 02:50

Mathematics, 08.05.2021 02:50



English, 08.05.2021 02:50