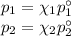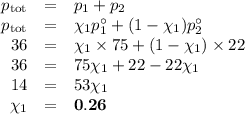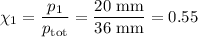Chemistry, 25.12.2019 03:31, vlactawhalm29
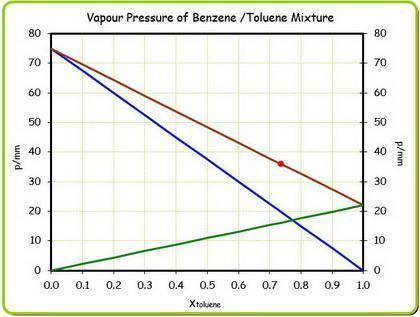
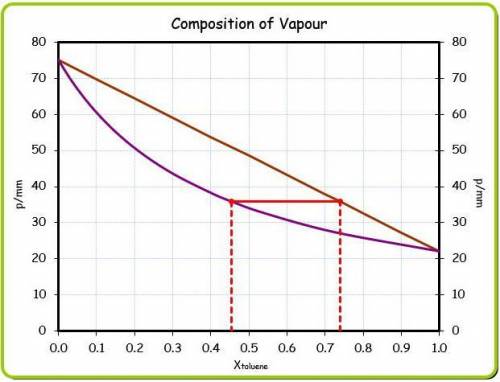
At 20 ? c the vapor pressure of benzene (c6h6)is 75 torr, and that of toluene (c7h8) is 22 torr
. assume that benzene and toluene form an ideal solution.
what is the composition in mole fractions of a solution that has a vapor pressure of 36torr at 20 degrees c?
what is the mole fraction of benzene in the vapor above the solution described in part (a)?

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 07:30, kimberlyrios12p0ts98
Which of the following best supports the concept that genetic information is passed on to offspring from both of their parents, not just one?
Answers: 2

Chemistry, 22.06.2019 10:40, yfgkeyonna
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2
Do you know the correct answer?
At 20 ? c the vapor pressure of benzene (c6h6)is 75 torr, and that of toluene (c7h8) is 22 torr
Questions in other subjects:



Advanced Placement (AP), 10.02.2021 06:20

Chemistry, 10.02.2021 06:20

English, 10.02.2021 06:20


Mathematics, 10.02.2021 06:20

Mathematics, 10.02.2021 06:20

English, 10.02.2021 06:20

Mathematics, 10.02.2021 06:20