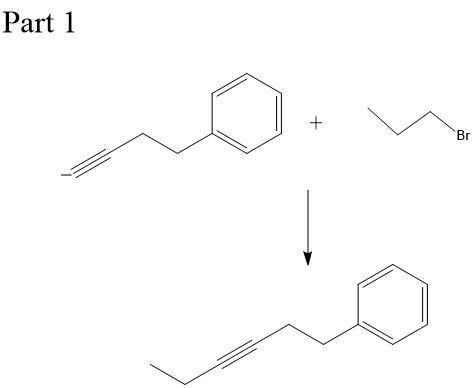
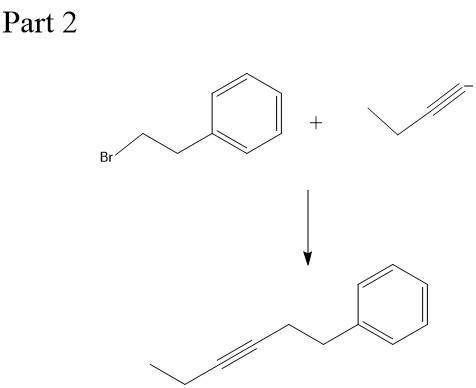
Chemistry, 23.12.2019 21:31, naomicervero
This molecule can be synthesized from an alkyne anion and an alkyl bromide. however, there are two ways in which this molecule can be formed. one way uses a higher molecular weight alkyne anion (part 1) and the other uses a lower molecular weight anion (part 2). draw the two versions in the boxes below. omit spectator ions.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, colochaortiz20p7cajw
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1


Chemistry, 23.06.2019 05:00, jjoyner
Question 5 match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) reactant that can produce more of the product theoretical yield c) amount of product predicted to be produced by the given reactants
Answers: 2

Chemistry, 23.06.2019 13:40, gtamods402
Which of the following volumes is the smallest? a) one microliter b)one deciliter d)one liter c)one milliliter
Answers: 2
Do you know the correct answer?
This molecule can be synthesized from an alkyne anion and an alkyl bromide. however, there are two w...
Questions in other subjects:


French, 02.06.2021 01:20

Mathematics, 02.06.2021 01:20




English, 02.06.2021 01:20