
Chemistry, 20.12.2019 23:31, michaelchavez6959127
In a different experiment, a student places a piece of pure ba(s) in a beaker containing 250.ml of 6.44mhcl(aq) and observes that the rate of appearance of h2(g) bubbles when ba(s) was added is much faster than the rate of appearance of bubbles when the mg(s) was added. the student claims that the greater reactivity is due to the difference in ionization energy between mg and ba. explain the difference in ionization energy between mg and ba in terms of atomic structure.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, VictoriaRose520
This is a characteristic of the elements in the periodic table that shows a pattern. it may increase or decrease across or down the table.
Answers: 1


Chemistry, 22.06.2019 04:20, lex68259100
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1
Do you know the correct answer?
In a different experiment, a student places a piece of pure ba(s) in a beaker containing 250.ml of 6...
Questions in other subjects:



Mathematics, 28.01.2020 04:31

Chemistry, 28.01.2020 04:31

Mathematics, 28.01.2020 04:31


Health, 28.01.2020 04:31




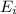
![[Ne]3s^2](/tpl/images/0428/3026/d3dcb.png)
![[Xe]6s^2](/tpl/images/0428/3026/f6f70.png)





