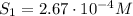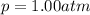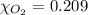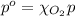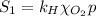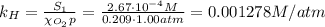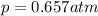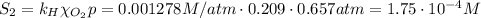The solubility of oxygen in lakes high in the rocky mountains is affected by the altitude. if the solubility of o2 from the air is 2.67 ✕ 10-4 m at sea level and 25°c, what is the solubility of o2 at an elevation of 12,000 ft where the atmospheric pressure is 0.657 atm? assume the temperature is 25°c, and that the mole fraction of o2 in air is 0.209 at both 12,000 ft and at sea level.

Answers: 2
Other questions on the subject: Chemistry



Do you know the correct answer?
The solubility of oxygen in lakes high in the rocky mountains is affected by the altitude. if the so...
Questions in other subjects:

Mathematics, 11.02.2021 20:40

Mathematics, 11.02.2021 20:40



Mathematics, 11.02.2021 20:40




Mathematics, 11.02.2021 20:40


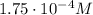
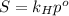
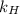 is Henry's law constant.
is Henry's law constant.