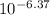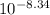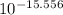Chemistry, 19.12.2019 05:31, aruhter2843
Astudent was given a 0.15 m solution of an unknown diprotic acid h2a and asked to determine the ka1 and ka2 values for the diprotic acid. the student titrated 46 ml of the 0.15 m h2a with 0.15 m koh. after 23.0 ml of koh was added, the ph of the resulting solution was 6.37. after 46 ml of koh was added, the ph of the resulting solution was 8.34. what are the values of ka1 and ka2 for the diprotic acid?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, only1cache
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3

Chemistry, 22.06.2019 05:30, ayoismeisjjjjuan
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 07:30, 10040813
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
Do you know the correct answer?
Astudent was given a 0.15 m solution of an unknown diprotic acid h2a and asked to determine the ka1...
Questions in other subjects:




Biology, 02.10.2020 15:01



Biology, 02.10.2020 15:01

Law, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01

Mathematics, 02.10.2020 15:01