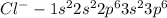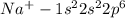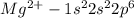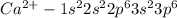Chemistry, 18.12.2019 23:31, 2020seogang
Write the electron configuration for the monatomic ions formed ions formed from the following elements (which form the greatest concentration of monatomic ions in seawater):
(a) cl
(b) na
(c)mg
(d) ca

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:30, noathequeen
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3


Chemistry, 22.06.2019 10:00, youngchapo813p8d9u1
Main expenses you plan on making payments on a new car too. you want to spend 15% of your monthly net pay on the car payment, insurance, registration, and taxes combined. what is your monthly car allowance? $149.46 $298.91 $448.37 $597.83
Answers: 3
Do you know the correct answer?
Write the electron configuration for the monatomic ions formed ions formed from the following elemen...
Questions in other subjects:

History, 14.02.2021 05:40


Mathematics, 14.02.2021 05:40


Health, 14.02.2021 05:40



Arts, 14.02.2021 05:40


History, 14.02.2021 05:40