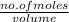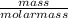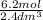What is the molarity of a 2.4-liter solution containing 124 grams of hf?
a. 2.6 m hf &l...

Chemistry, 09.01.2020 09:31, klaytminecraft
What is the molarity of a 2.4-liter solution containing 124 grams of hf?
a. 2.6 m hf < my guess based on calculations
b. 6.2 m hf
c. 8.3 m hf
d. 15 m hf

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:30, amandajbrewerdavis
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 13:30, citlalli30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 22.06.2019 13:30, kkingstone7062
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Chemistry, 22.06.2019 15:30, lizzyhearts
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3
Do you know the correct answer?
Questions in other subjects:

Mathematics, 14.11.2019 21:31

English, 14.11.2019 21:31



English, 14.11.2019 21:31


Mathematics, 14.11.2019 21:31



Mathematics, 14.11.2019 21:31