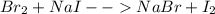Unknown halogen x2 was added to each of two test tubes which contained aqueous nacl and aqueous nai, respectively. when hexane was added to the test tubes, an orange color developed in the top layer of the first test tube, while purple developed in the second.
a. identify x2.
b. write a balanced equation for each reaction which occurred.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 19:00, georgesarkes12
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2
Do you know the correct answer?
Unknown halogen x2 was added to each of two test tubes which contained aqueous nacl and aqueous nai,...
Questions in other subjects:


English, 17.01.2020 17:31






Biology, 17.01.2020 17:31

Mathematics, 17.01.2020 17:31


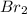
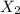 is
is 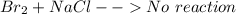 (because bromine is less reactive than chlorine)
(because bromine is less reactive than chlorine)