Chemistry, 18.12.2019 02:31, gwendallinesikes
Calculate the change in internal energy (δe) for a system that is absorbing 35.8 kj of heat and is expanding from 8.00 to 24.0 l in volume at 1.00 atm. (remember that 101.3 j = 1 l·atm)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, AbhiramAkella
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 15:30, alaf05160
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks. energy was destroyed inside the blocks. energy was absorbed into the blocks from outside the system. energy was transferred from the warmer block to the cooler block.
Answers: 2
Do you know the correct answer?
Calculate the change in internal energy (δe) for a system that is absorbing 35.8 kj of heat and is e...
Questions in other subjects:




Physics, 04.02.2021 20:10

Mathematics, 04.02.2021 20:10

Computers and Technology, 04.02.2021 20:10



Mathematics, 04.02.2021 20:10


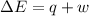
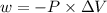
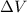 is the change in volume
is the change in volume
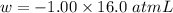
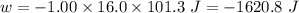 (work is done by the system)
(work is done by the system)