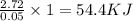Chemistry, 18.12.2019 01:31, mallorynichole19
When a student mixes 50 ml of 1.0 m hcl and 50 ml of 1.0 m naoh in a coffee-cup calorimeter, the temperature of the resultant solution increases from 21.0 °c to 27.5 °c. calculate the enthalpy change for the reaction in kj per mol of hcl, assuming that the calorimeter loses only a negligible quantity of heat. the total volume of the solution is 100 ml, its density is 1.0 g/ml, and its specific heat is 4.18 j/g*k.

Answers: 2
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 21:00, cxttiemsp021
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
Do you know the correct answer?
When a student mixes 50 ml of 1.0 m hcl and 50 ml of 1.0 m naoh in a coffee-cup calorimeter, the tem...
Questions in other subjects:


Mathematics, 08.10.2020 04:01

Mathematics, 08.10.2020 04:01

Geography, 08.10.2020 04:01




Computers and Technology, 08.10.2020 04:01



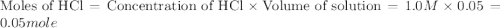
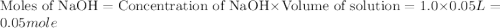

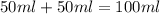
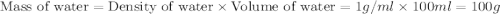
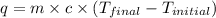
 = specific heat of water =
= specific heat of water = 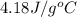
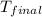 = final temperature of water =
= final temperature of water = 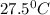
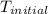 = initial temperature of metal =
= initial temperature of metal = 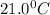
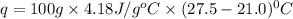
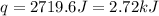
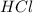 :
: