
Consider the following reaction:
2hcl(g) + 185 kj → h2(g) + cl2(g)
describe the change in energy according to the law of conservation of energy.
a) there is a net gain of 185 kj of energy when decomposing hcl.
b) it takes more energy to break the bonds of hcl than form h2(g) + cl2(g).
c) the energy required to decompose hcl and produce h2(g) + cl2(g) are equal.
d) it takes less energy to break the bonds of hcl than form the bonds of h2(g) + cl2(g.)

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:20, blondielocks2002
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3

Chemistry, 22.06.2019 15:40, alleshia2007
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 19:20, evansh78
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
Do you know the correct answer?
Consider the following reaction:
2hcl(g) + 185 kj → h2(g) + cl2(g)
describe the c...
2hcl(g) + 185 kj → h2(g) + cl2(g)
describe the c...
Questions in other subjects:


Physics, 07.10.2019 09:10




Mathematics, 07.10.2019 09:10


English, 07.10.2019 09:10


Geography, 07.10.2019 09:10


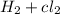 are equal.
are equal.



