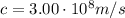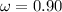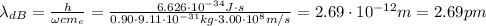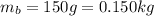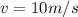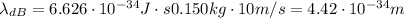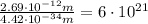Chemistry, 17.12.2019 07:31, ddwithdadarco
Calculate "de broglie" wavelength for each of the following, and use your numerical answers the to explain why macroscopic (large) objects are not ordinarily discussed in terms of their "wave-like" properties. a. an electron moving at .90 times the speed of light.
b. a 150-g ball moving at a speed of 10.m/s

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, micvar9646
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 07:30, 10040813
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 09:00, SilverTheAmarok
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2

Chemistry, 22.06.2019 09:40, cheesecake1919
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3
Do you know the correct answer?
Calculate "de broglie" wavelength for each of the following, and use your numerical answers the to e...
Questions in other subjects:

Mathematics, 13.01.2021 08:30

Mathematics, 13.01.2021 08:30


Mathematics, 13.01.2021 08:30


Computers and Technology, 13.01.2021 08:30

Mathematics, 13.01.2021 08:30


Mathematics, 13.01.2021 08:30

Chemistry, 13.01.2021 08:30


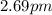
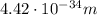
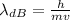
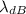 is the de Broglie wavelength (in m);
is the de Broglie wavelength (in m);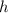 is the Planck's constant,
is the Planck's constant, 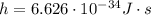 ;
;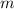 is mass (in kg);
is mass (in kg);![v[tex] is velocity (in m/s).a. We need to know the mass of an electron here:[tex]m_e=9.11\cdot10^{-31} kg](/tpl/images/0422/1849/0b8f1.png)