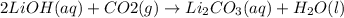The carbon dioxide exhaled by astronauts can be removed by its reaction with lithium hydroxide, lioh, according to the following chemical equation: co2(g) + 2lioh(s) — li2co3(s)
+ h2o4). how many moles of lithium carbonate are produced when 20.0 mol co2 the average
amount exhaled by a person each day, reacts completely with lithium hydroxide?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:40, yfgkeyonna
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2


Chemistry, 22.06.2019 18:50, emily9656
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3
Do you know the correct answer?
The carbon dioxide exhaled by astronauts can be removed by its reaction with lithium hydroxide, lioh...
Questions in other subjects:



Mathematics, 23.06.2020 00:57





History, 23.06.2020 10:57


English, 23.06.2020 10:57