Which substances have ah = 0 kj/mol
...

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:30, emmalybrown
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Mathematics, 27.09.2020 14:01

Chemistry, 27.09.2020 14:01



Mathematics, 27.09.2020 14:01




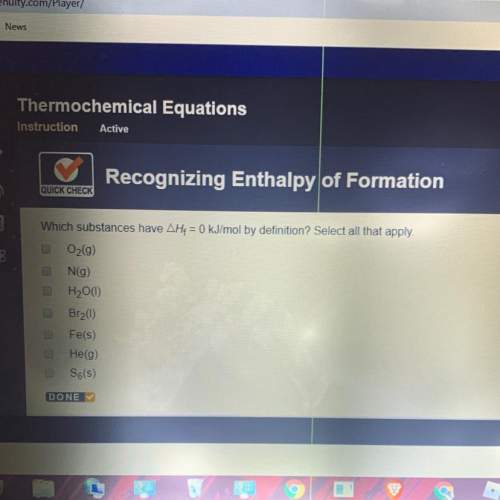
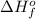 is defined as the standard enthalpy of formation. By definition, the standard enthalpy of formation is equal to 0 kJ/mol for the substances in their standard states, that is, at room temperature and 1 atm pressure.
is defined as the standard enthalpy of formation. By definition, the standard enthalpy of formation is equal to 0 kJ/mol for the substances in their standard states, that is, at room temperature and 1 atm pressure.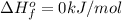 .Although nitrogen is a gas at standard conditions, it is diatomic, so
.Although nitrogen is a gas at standard conditions, it is diatomic, so 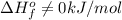 .Water is a liquid at standard conditions, however, it consists of two types of atoms, hydrogen and oxygen, so
.Water is a liquid at standard conditions, however, it consists of two types of atoms, hydrogen and oxygen, so 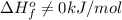 .Bromine is a liquid at standard conditions, so
.Bromine is a liquid at standard conditions, so 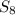 and not
and not 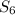 , so
, so 



