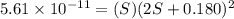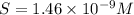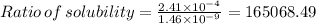Chemistry, 17.12.2019 02:31, winterblanco
Mg(oh)2 is a sparingly soluble salt with a solubility product, ksp, of 5.61×10−11. it is used to control the ph and provide nutrients in the biological (microbial) treatment of municipal wastewater streams. what is the ratio of solubility of mg(oh)2 dissolved in pure h2o to mg(oh)2 dissolved in a 0.180 m naoh solution? express your answer numerically as the ratio of molar solubility in h2o to the molar solubility in naoh.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, jamesnaquan132
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 14:00, emilyproce
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 23.06.2019 00:50, maddysmall32
Which of the following warnings would an agricultural chemist tell a farmer who wants to recycle his or her own ammonia? recycling ammonia is a difficult process that sometimes takes weeks. recycling ammonia requires a degree in biochemistry or a related field. recycling ammonia can be harmful because it is highly flammable and toxic. recycling ammonia costs too much money considering the price of the necessary chemicals.
Answers: 1

Chemistry, 23.06.2019 07:20, msladycie8831
F1.5 mol of nabh4 react, how many moles of b2h6 are formed? 2 nabh4(aq) + h2so4(aq) → 2 h2(g) + na2so4(aq) + b2h6(g)
Answers: 1
Do you know the correct answer?
Mg(oh)2 is a sparingly soluble salt with a solubility product, ksp, of 5.61×10−11. it is used to con...
Questions in other subjects:








Mathematics, 21.08.2019 17:30



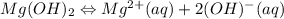
![[Mg^{2+}][OH^{-}]](/tpl/images/0421/6722/fd650.png)
![K_{sp}=[Mg^{2+}][OH^{-}]](/tpl/images/0421/6722/17c44.png)
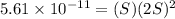
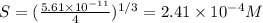
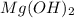 in 0.180 M NaOH is a follows.
in 0.180 M NaOH is a follows.