
Chemistry, 17.12.2019 01:31, Thejollyhellhound20
Consider the following general voltaic cell and a cell notation, and answer all three parts of this question.
mg(s) | mg2+(aq) || cl2(g) | cl−(aq) | c(s)
part 1: based on the cell notation, the substance of electrode a is [ select ] ["mg(s)", "c(s)", ""] , and the substance of electrode b is [ select ] ["c(s)", "mg(s)"] .
part 2: what is the balanced redox equation for the voltaic cell?
a) mg2+(aq) + 2 cl−(aq) \rightarrow → mg(s) + cl2(g)
b) mg2+(aq) + cl−(aq) \rightarrow → mg(s) + cl2(g)
c) mg(s) + cl2(g) \rightarrow → mg2+(aq) + 2 cl−(aq)
d) mg(s) + cl2(g) \rightarrow → mg2+(aq) + cl−(aq)
e) none of the above is correct, because c(s) doesn't appear in any of them.
part 3:
- if the salt bridge contains nano3, na+ ions flow to the [ select ] ["left", "right"] , and no3− ions flow to the [ select ] ["left", "right"] .
- electrons flow from electrode [ select ] ["a", "b"] to electrode [ select ] ["b", "a"] .

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 21:30, emmalucilleblaha1995
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
Do you know the correct answer?
Consider the following general voltaic cell and a cell notation, and answer all three parts of this...
Questions in other subjects:

Mathematics, 04.05.2020 23:42


History, 04.05.2020 23:42

History, 04.05.2020 23:42


Mathematics, 04.05.2020 23:42





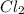
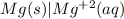
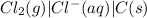
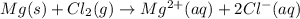
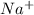 ions flow from the left to right.
ions flow from the left to right.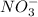 ions flow from the right to left.
ions flow from the right to left.



