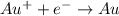Chemistry, 16.12.2019 19:31, Aliyah2020
Acell consists of a gold wire and a saturated calomel electrode (s. c.e.) in a 0.130 m auno 3 solution at 25 °c. the gold wire is connected to the positive end of a potentiometer, and the s. c.e. is connected to the negative end of the potentiometer. what is the half‑reaction that occurs at the au electrode? include physical states.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:20, shanyeah
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1


Chemistry, 22.06.2019 22:30, teagan56
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1
Do you know the correct answer?
Acell consists of a gold wire and a saturated calomel electrode (s. c.e.) in a 0.130 m auno 3 soluti...
Questions in other subjects:

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Biology, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Geography, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01