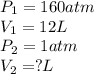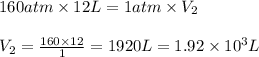According to boyle's law, for a fixed quantity of gas at a given temperature, what quantity relating pressure p and volume v is constant? view available hint(s) according to boyle's law, for a fixed quantity of gas at a given temperature, what quantity relating pressure and volume is constant?
a. pv
b. p×v
c. p+v
d. vp
application of boyle's law a 12-liter tank contains helium gas pressurized to 160 atm.
part b what size tank would be needed to contain this same amount of helium at atmospheric pressure (1 atm)? express the size in liters to three significant figures. view available hint(s) nothing ll

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:00, kingamir
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
Do you know the correct answer?
According to boyle's law, for a fixed quantity of gas at a given temperature, what quantity relating...
Questions in other subjects:



History, 08.04.2020 03:30





English, 08.04.2020 03:30


English, 08.04.2020 03:30


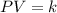 and the size of the tank must be
and the size of the tank must be 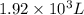
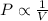
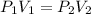
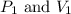 are initial pressure and volume.
are initial pressure and volume.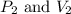 are final pressure and volume.
are final pressure and volume.