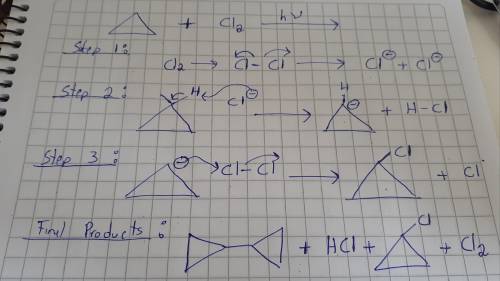
Be sure to answer all parts. cyclopropyl chloride has been prepared by the free-radical chlorination of cyclopropane. draw the products for each equation of the stepwise mechanism for this reaction. be sure to include lone pair(s) of electrons where necessary. part 1 out of 3 ca45304fix1 draw structure plussign

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2



Do you know the correct answer?
Be sure to answer all parts. cyclopropyl chloride has been prepared by the free-radical chlorination...
Questions in other subjects:

Mathematics, 15.11.2020 20:00


Mathematics, 15.11.2020 20:00