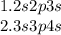Will mark
the complete orbital notation diagram of an atom is shown.
five squares are shown aligned horizontally. inside the first square from the left is shown one upwards pointing arrow and one downwards pointing arrow. in the second, third, and fourth squares is a pair of upwards and downwards pointing arrows. the fifth square has a single upwards pointing arrow.
based on the diagram, what values can be assigned to the principal quantum number for the electrons in the atom? what information does this quantum number provide about the location of the electron?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, tifftiff22
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2

Chemistry, 22.06.2019 08:30, omoaye
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 10:00, berniceallonce22
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1
Do you know the correct answer?
Will mark
the complete orbital notation diagram of an atom is shown.
five squares...
the complete orbital notation diagram of an atom is shown.
five squares...
Questions in other subjects:

Mathematics, 08.04.2021 17:10

Mathematics, 08.04.2021 17:10



History, 08.04.2021 17:10



Mathematics, 08.04.2021 17:10



 which is possible in two cases :
which is possible in two cases :