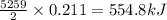Chemistry, 12.12.2019 06:31, alexdziob01
)b5h9(l) is a colorless liquid that will explode when exposed to oxygen. how much heat is released when 0.211 mol of b5h9 reacts with excess oxygen where the products are b2o3(s) and h2o(l). the standard enthalpy of formation of b5h9(l) is 73.2 kj/mol, the standard enthalpy of formation of b2o3(s) is -1272 kj/mol and that of h2o(l) is -285.4 kj/mol. express your answer in kj.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 05:00, jayden6467
How many moles are in 7.2 x 10^23 carbon molecules?
Answers: 1
Do you know the correct answer?
)b5h9(l) is a colorless liquid that will explode when exposed to oxygen. how much heat is released w...
Questions in other subjects:

Mathematics, 02.02.2021 03:30

Mathematics, 02.02.2021 03:30

Mathematics, 02.02.2021 03:30

Mathematics, 02.02.2021 03:30





Spanish, 02.02.2021 03:30

Mathematics, 02.02.2021 03:30


 reacts is 554.8 kJ
reacts is 554.8 kJ with oxygen gas follows:
with oxygen gas follows: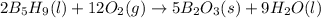
![\Delta H_{rxn}=[(5\times \Delta H_f_{(B_2O_3(s))})+(9\times \Delta H_f_{(H_2O(l))})]-[(2\times \Delta H_f_{(B_5H_9(l))})+(12\times \Delta H_f_{(O_2(g))})]](/tpl/images/0414/9485/05a9d.png)
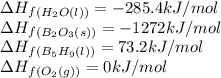
![\Delta H_{rxn}=[(2\times (-1272))+(9\times (-285.4))]-[(2\times (73.2))+(12\times (0))]\\\\\Delta H_{rxn}=-5259kJ](/tpl/images/0414/9485/eaac9.png)