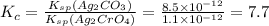Chemistry, 11.12.2019 19:31, mariahrpoulin1511
The value of the solubility product constant for ag2co3 is 8.5 × 10‒12 and that of ag2cro4 is 1.1 × 10‒12. from this data, what is the value of kc for the reaction, ag2co3(s) + cro42‒(aq) → ag2cro4(s) + co32‒(aq) a) 9.6 × 10‒12 b) 7.7 c) 1.1 × 1023 d) 1.3 × 10‒1 e) 9.4 × 10‒24

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, sotoamerica0814
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 03:30, jabper5522
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1


Chemistry, 22.06.2019 12:30, johnsont8377
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1
Do you know the correct answer?
The value of the solubility product constant for ag2co3 is 8.5 × 10‒12 and that of ag2cro4 is 1.1 ×...
Questions in other subjects:

Mathematics, 07.10.2020 07:01

Biology, 07.10.2020 07:01

Mathematics, 07.10.2020 07:01


English, 07.10.2020 07:01


Mathematics, 07.10.2020 07:01

Biology, 07.10.2020 07:01



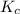 for the given reaction is 7.7
for the given reaction is 7.7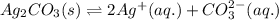
![K_{sp}(Ag_{2}CO_{3})=[Ag^{+}]^{2}[CO_{3}^{2-}]](/tpl/images/0413/8617/b8832.png)
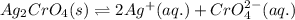
![K_{sp}(Ag_{2}CrO_{4})=[Ag^{+}]^{2}[CrO_{4}^{2-}]](/tpl/images/0413/8617/31f53.png)
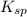 represents solubility product
represents solubility product![K_{c}=\frac{[CO_{3}^{2-}]}{[CrO_{4}^{2-}]}](/tpl/images/0413/8617/fea9e.png) (concentration of pure solids remain constant during reaction. Hence their concentration is taken as 1 to exclude them from equilibrium constant expression)
(concentration of pure solids remain constant during reaction. Hence their concentration is taken as 1 to exclude them from equilibrium constant expression)![K_{c}=\frac{[Ag^{+}]^{2}[CO_{3}^{2-}]}{[Ag^{+}]^{2}[CrO_{4}^{2-}]}](/tpl/images/0413/8617/3fc1b.png)