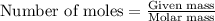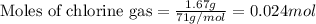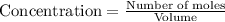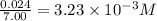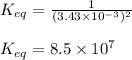Titanium and chlorine react to form titanium(iv) chloride, like this: ti(s) + 2 cl 2(g)-ticl 4( at a certain temperature, a chemist finds that a 7.0 l reaction vessel containing a mixture of titanium, chlorine, and titanium(iv) chloride at equilibrium has the following composition compound amount 1.67 g cl 2.93 g tici 2.02 g ti calculate the value of the equilibrium constant k for this reaction. round your answer to 2 significant digits.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:30, tylerineedhelp
Ihat will happen if i added baking soda to vinegar
Answers: 2

Chemistry, 22.06.2019 05:20, barry14201
Identify and describe the three ways that mutations affect organisms.
Answers: 1
Do you know the correct answer?
Titanium and chlorine react to form titanium(iv) chloride, like this: ti(s) + 2 cl 2(g)-ticl 4( at...
Questions in other subjects:

Mathematics, 17.08.2020 21:01

English, 17.08.2020 21:01






Mathematics, 17.08.2020 21:01

Health, 17.08.2020 21:01

Mathematics, 17.08.2020 21:01



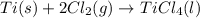
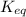 for the above reaction follows:
for the above reaction follows:![K_{eq}=\frac{1}{[Cl_2]^2}](/tpl/images/0413/8706/8ac7b.png)