
Consider two flasks at 25 degree celsius, one contains an ideal gas and the other contains the real gas so3. which statement regarding these gases is true?
a. as the temperature approaches 0 k, the volume of the ideal gas will be larger than the volume of so3 because ideal gases lack inter-molecular forces.
b. as the temperature is decreased, the ideal gas will liquefy first because ideal gases have larger values of the van der waals coefficient b.
c. as the temperature approaches 0 k, the volume of the ideal gas will be smaller than the volume of so3 because ideal gases have larger values of the van der waals coefficient a.
d. after the temperature has been lowered about 100 k, the pressure of the ideal gas will be smaller than the pressure of so3 because the van der waals coefficient a for so3 is large.
e. after the temperature is increased about 100 k, the pressure of the ideal gas will be smaller than the pressure of so3 because the van der waals coefficient a for so3 is lar

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:10, GreatBaconGamer
Which class of molecules functions as chemical signals? hormones water carbohydrates proteins
Answers: 1

Chemistry, 22.06.2019 12:30, MrSavannahCat
Clyde and marilyn are riding a roller coaster. during which section(s) of the track is their potential energy converted to kinetic energy? a. from point b to point c only b. from point b to point d only c. from point a to point b only d. from point a to point b and from point c to point d
Answers: 1

Chemistry, 23.06.2019 00:30, zaniathomasel
What is bromine+calcium iodide--> calcium bromide +iodine balanced
Answers: 1
Do you know the correct answer?
Consider two flasks at 25 degree celsius, one contains an ideal gas and the other contains the real...
Questions in other subjects:

Mathematics, 22.07.2021 14:00


English, 22.07.2021 14:00





Social Studies, 22.07.2021 14:00


Mathematics, 22.07.2021 14:00


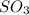 because ideal gases lack inter-molecular forces, is true.
because ideal gases lack inter-molecular forces, is true.



