
Chemistry, 09.12.2019 17:31, bartekzelazek5083
X+ 2y > xy2in order to determine the order of the reaction represented above, the initial rate of formation of xy2 is measured using different initial values of [x] and [y]. the results of the experiment are shown in the table below. trial [x] [y] rate of formation [xy2]1 .5 m .5 m 8.0 x 10^-3 m/s2 1.0 m .5 m 3.2 x 10^-2 m/s3 1.0 m 1.0 m 6.4 x 10^-2 m/sin trial 2 which of the reactants would be consumed more rapidly and why? x, because the reaction is second order with respect to x. y, because the reaction is second order with respect to y. x, because it has a higher molar concentration. y, because the rate of disappearance will be double that of x.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, 5041
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1

Chemistry, 21.06.2019 22:00, toledanomariap43bxm
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3


Chemistry, 22.06.2019 14:00, coylenoah0
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2
Do you know the correct answer?
X+ 2y > xy2in order to determine the order of the reaction represented above, the initial rate o...
Questions in other subjects:

History, 11.01.2020 23:31

Biology, 11.01.2020 23:31



Mathematics, 11.01.2020 23:31




Mathematics, 11.01.2020 23:31


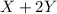 ⇒
⇒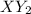
![\left[\begin{array}{ccc}[X]&[Y]&rate\\1.5M&0.5M&8*10^{-3}Ms^{-1}\\21M&0.5M&3.2*10^{-2}Ms^{-1}\\31M&1M&6.4*10^{-2}Ms{-1}\end{array}\right]](/tpl/images/0410/1221/efcd6.png)
 is ,
is ,![r \alpha [X]^a[Y]^b](/tpl/images/0410/1221/1f54d.png)
 and
and 
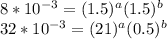
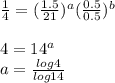
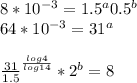 a=0.5253b=0.7048
a=0.5253b=0.7048




