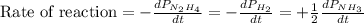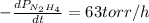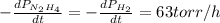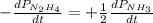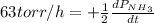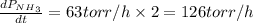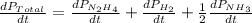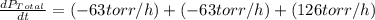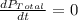Chemistry, 07.12.2019 00:31, kaylarenee05080
The rate of decrease in n2h4 partial pressure in a closed reaction vessel form the reaction: n2h4 (g) + h2 (g) \rightarrow 2 nh3 (g) is63 torr/h. what are the rates of change of nh3 partial pressure and total pressure in the vessel.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:10, yootmytoot
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution. calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1


Chemistry, 23.06.2019 04:31, woodfordmaliky
One student said that the investigation was not valid (a fair test). write a plan for the investigation that includes improvements to the method and apparatus
Answers: 1

Chemistry, 23.06.2019 08:30, aambitiouss
Of element x has 22 protons, how many electrons does it have
Answers: 1
Do you know the correct answer?
The rate of decrease in n2h4 partial pressure in a closed reaction vessel form the reaction: n2h4 (...
Questions in other subjects:

English, 26.08.2019 20:50

Mathematics, 26.08.2019 20:50

Mathematics, 26.08.2019 20:50




Mathematics, 26.08.2019 20:50



French, 26.08.2019 20:50


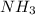 partial pressure is 126 torr/h.
partial pressure is 126 torr/h.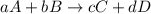
![\text{Rate of disappearance of A}=-\frac{1}{a}\frac{d[A]}{dt}](/tpl/images/0407/2122/2d8eb.png)
![\text{Rate of disappearance of B}=-\frac{1}{b}\frac{d[B]}{dt}](/tpl/images/0407/2122/1e77e.png)
![\text{Rate of formation of C}=+\frac{1}{c}\frac{d[C]}{dt}](/tpl/images/0407/2122/cee4b.png)
![\text{Rate of formation of D}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0407/2122/7ef32.png)
![Rate=-\frac{1}{a}\frac{d[A]}{dt}=-\frac{1}{b}\frac{d[B]}{dt}=+\frac{1}{c}\frac{d[C]}{dt}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0407/2122/d4b94.png)
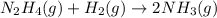
![\text{Rate of disappearance of }N_2H_4=-\frac{d[N_2H_4]}{dt}](/tpl/images/0407/2122/4b6d3.png)
![\text{Rate of disappearance of }H_2=-\frac{d[H_2]}{dt}](/tpl/images/0407/2122/53b46.png)
![\text{Rate of formation of }NH_3=+\frac{1}{2}\frac{d[NH_3]}{dt}](/tpl/images/0407/2122/f55ec.png)