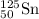Chemistry, 06.12.2019 00:31, ninilizovtskt
An atom of 125sn has a mass of 124.907785 amu. calculate the mass defect (deficit) in amu/atom. use the masses: mass of 1h atom = 1.007825 amu mass of a neutron = 1.008665 amu 1 amu = 931.5 mev give your answer to 4 significant figures and do not use e notation..

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, webbhlharryteach
In which of these cases are the two wave points considered to be in phase with each other?
Answers: 1


Chemistry, 22.06.2019 13:00, rome58
Lab reagent, hypothesis test. a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl. these six measurements are assumed to be an srs of all possible measurements from solution. they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution. carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1

Chemistry, 22.06.2019 15:30, lovebaeforlife351
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins. co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1
Do you know the correct answer?
An atom of 125sn has a mass of 124.907785 amu. calculate the mass defect (deficit) in amu/atom. use...
Questions in other subjects:

Mathematics, 21.04.2020 08:21




Mathematics, 21.04.2020 08:21

Health, 21.04.2020 08:21


Mathematics, 21.04.2020 08:21

English, 21.04.2020 08:21

Mathematics, 21.04.2020 08:21