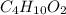Chemistry, 05.12.2019 05:31, annemcnair217
What is the empirical formula of a compound with the molecular formula of c4h10o2?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:50, kelli151
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2

Chemistry, 22.06.2019 15:00, tcapele252
‘which reaction would most likely require the use of an inert electrode?
Answers: 1


Chemistry, 23.06.2019 04:20, tyrickdavis1
The equation below shows a chemical reaction. a + b + heat —> c + d according to the law of conservation of energy, which statement is true? a. the reactants absorb heat because they have less energy than the products. b. the products release heat because they have more energy than the reactants. c. the reactants generate heat because they have more energy than the products. d. the products require heat to form because they have less energy than the reactants.
Answers: 1
Do you know the correct answer?
What is the empirical formula of a compound with the molecular formula of c4h10o2?...
Questions in other subjects:

Mathematics, 08.07.2019 15:00




Biology, 08.07.2019 15:00

Health, 08.07.2019 15:00





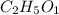 .
.