Chemistry, 05.12.2019 01:31, billythe1st
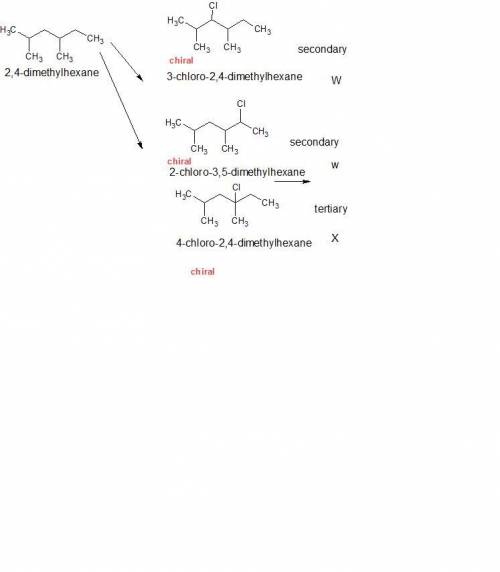
Compound w, c8h17br, and x, c8h17cl, are products of the radical bromination (w), and chlorination (x), of 2,4-dimethylhexane. w and x are each chiral. w reacts readily in water to form a chiral substitution product y, c8h17oh. reaction of a single enantiomer of w produces a single enantiomer of y as the only substitution product. x likewise reacts readily in water though at a slower rate than w; a chiral substitution product z, c8h17oh, is again formed. unlike w, however, reaction of a single enantiomer of x produces z that is nearly racemic. propose structures for w and x.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:20, mgavyn1052
Calculate the molarity of the solution. 6.02 x 1022 molecules of hci (molecular weight = 36.5 g/mole) in 2.0 liters of water m
Answers: 1

Chemistry, 22.06.2019 05:30, livigrace9004
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2


Chemistry, 22.06.2019 15:30, abdullaketbi71
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1
Do you know the correct answer?
Compound w, c8h17br, and x, c8h17cl, are products of the radical bromination (w), and chlorination (...
Questions in other subjects:


Mathematics, 12.01.2021 19:20



English, 12.01.2021 19:20