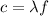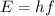Chemistry, 03.12.2019 06:31, genyjoannerubiera
Fireworks that contain metallic salts such as sodium, strontium, and barium can generate bright colors. a technician investigates what colors are produced by the metallic salts by performing flame tests. during a flame test, a metallic salt is heated in the flame of a gas burner. each metallic salt emits a characteristic colored light in the flame. 3. state how bright-line spectra viewed through a spectroscope can be used to identify the metal ions in the salts used in the flame tests. 4. explain, in terms of electrons, how a strontium salt emits colored light. 5. explain why the electron configuration of 2-7-1-1 represents a sodium atom in an excited state.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, AnastasiaJauregui
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1

Chemistry, 22.06.2019 15:40, alleshia2007
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 23.06.2019 00:00, PlzNoToxicBan
What is the pressure of 0.500 moles of carbon dioxide gas in a 2.5 l tank and at a temperature of 301 k? (r=0.0821 l·atm/mol·k) 3.08 atm 1.2 atm 0.23 atm 4.01 atm 4.94 atm
Answers: 1

Chemistry, 23.06.2019 02:00, xoxoadara13ox07ck
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1
Do you know the correct answer?
Fireworks that contain metallic salts such as sodium, strontium, and barium can generate bright colo...
Questions in other subjects:

English, 23.09.2019 10:30



Mathematics, 23.09.2019 10:30


Mathematics, 23.09.2019 10:30

History, 23.09.2019 10:30

Mathematics, 23.09.2019 10:30