
Chemistry, 03.12.2019 05:31, summerjoiner
Asample of 0.495 grams of solid khp is weighed into an erlenmeyer flask. this sample is titrated with a sodium hydroxide solution, and 28.56 ml of naoh are required to reach the endpoint. the sodium hydroxide solution is then used to titrate a sample of phosphoric acid of unknown concentration. it requires 29.88 ml of naoh to react with 10.33 ml of h3po4 solution. what is the concentration of the phosphoric acid?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, KindaSmartPersonn
What is the formula for the molecular compound nitrogen monoxide
Answers: 1

Chemistry, 22.06.2019 15:00, alanmarcus22
What does the symbol (–hfus) indicate in a phase change?
Answers: 1

Do you know the correct answer?
Asample of 0.495 grams of solid khp is weighed into an erlenmeyer flask. this sample is titrated wit...
Questions in other subjects:


Mathematics, 04.05.2021 04:00





Mathematics, 04.05.2021 04:00


Social Studies, 04.05.2021 04:00


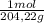 = 2,42x10⁻³ moles of KHP
= 2,42x10⁻³ moles of KHP




