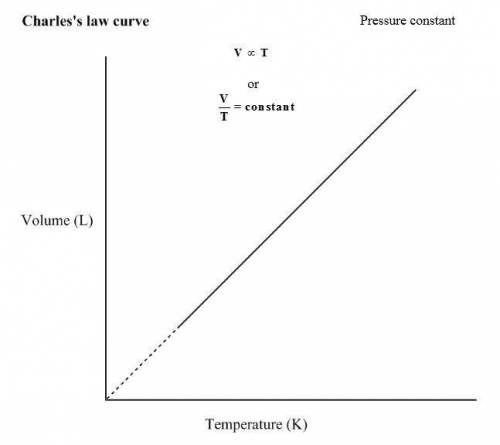
Charles's Law states that the volume and absolute temperature of a fixed quantity of gas are directly proportional under constant pressure conditions
Further explanation
There are several gas equations in various processes:
1. The general ideal gas equation
PV = nRT
PV = NkT
N = number of gas particles
n = number of moles
R = gas constant (8,31.10 ^ 3 J / kmole K
k = Boltzmann constant (1,38.10 ^ -23)
n = = N / No
n = m / M
n = mole
No = Avogadro number (6.02.10 ^ 23)
m = mass
M = relative molecular mass
2. Avogadro's hypothesis
In the same temperature and pressure, in the same volume conditions, the gas contains the same number of molecules
So it applies: the ratio of gas volume will be equal to the ratio of gas moles
V1: V2 = n1: n2
2. Boyle's Law
At a fixed temperature, the gas volume is inversely proportional to the pressure applied
p1.V1 = p2.V2
3. Charles's Law
When the gas pressure is kept constant, the gas volume is proportional to the temperature
V1 / T1 = V2 / T2
4. Gay Lussac's Law
When the gas is heated in a tube whose volume does not change, the gas pressure in the tube is proportional to its absolute temperature
P1 / T1 = P2 / T2
5. Law of Boyle-Gay-Lussac
Combined with Boyle's law and Gay Lussac's law
P1.V1 / T1 = P2.V2 / T2
P1 = initial gas pressure (N / m2 or Pa)
V1 = initial gas volume (m3)
P2 = gas end pressure
V2 = the final volume of gas
T1 = initial gas temperature (K)
T2 = gas end temperature
So the correct answer is Charles' Law, where at constant pressure, the volume of gas will be inversely proportional to its temperature
Learn more
Identify all of the gas law equations that relate to the ideal gas law
link
the ideal gas law
link
equation agrees with the ideal gas law
link


















 states that volume occupied by a fixed quantity of a gas is directly proportional to the absolute temperature (Kelvin) at constant pressure.
states that volume occupied by a fixed quantity of a gas is directly proportional to the absolute temperature (Kelvin) at constant pressure.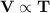 [P and n are constant]
[P and n are constant] [P and n are constant]
[P and n are constant]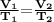 [P and n are constant]
[P and n are constant]