Chemistry, 29.11.2019 01:31, jessicapbailey52
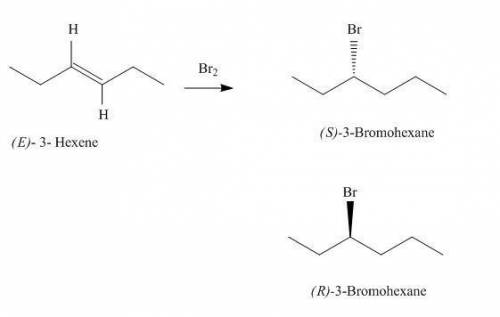
Be sure to answer all parts. show the overall reaction for formation of racemic 3−bromohexane from (e)−3−hexene by entering the structures of the products and reactants in the template provided. part 1 out of 3 draw structure arr draw structure + draw structure (e)−3−hexene (s)−3−bromohexane (r)−3−bromohexane racemic product = 50% (s) and 50% (r)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, mosthatedpicky1
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 08:30, breannaking9734
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 14:30, clemsongirl5392
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 17:50, kaylamount
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2
Do you know the correct answer?
Be sure to answer all parts. show the overall reaction for formation of racemic 3−bromohexane from (...
Questions in other subjects:



Mathematics, 24.05.2021 17:30



English, 24.05.2021 17:30

Mathematics, 24.05.2021 17:30

Social Studies, 24.05.2021 17:30


Business, 24.05.2021 17:30