
Chemistry, 28.11.2019 21:31, mervindavisk
Which of the following statements about the kinetic molecular theory are not correct?
a. the combined volume of all the molecules of the gas is negligible relative to the total volume in which the gas is contained
b. gases consist of molecules that are in continuous, random motion
c. the average kinetic energy of the molecules is not related to the absolute temperature
d. attractive and repulsive forces between gas molecules are negligible

Answers: 3
Other questions on the subject: Chemistry



Do you know the correct answer?
Which of the following statements about the kinetic molecular theory are not correct?
a. the...
a. the...
Questions in other subjects:



History, 11.10.2020 19:01

English, 11.10.2020 19:01


Mathematics, 11.10.2020 19:01




Biology, 11.10.2020 19:01


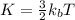
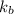 is the Boltzmann constant
is the Boltzmann constant



