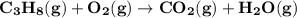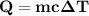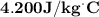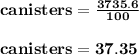On a 7 day wilderness expedition you'll need to heat 4.0kg of water to the boiling point each day. the air temperature will average 25°c. you have available canisters of compressed propane c3h8 fuel, which you'll burn to heat the water. what is the minimum number of fuel canisters you must bring?
the standard heat of formation of propane at 25°c is −103.85/kjmol.

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:40, Maryjasmine8001
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 05:50, Makoshark6887
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 06:40, CylieTbh
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3
Do you know the correct answer?
On a 7 day wilderness expedition you'll need to heat 4.0kg of water to the boiling point each day. t...
Questions in other subjects:

Social Studies, 01.04.2021 18:20

Mathematics, 01.04.2021 18:20

Advanced Placement (AP), 01.04.2021 18:20


History, 01.04.2021 18:20

Mathematics, 01.04.2021 18:20




English, 01.04.2021 18:30