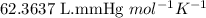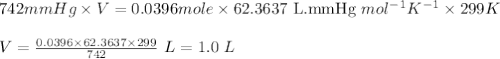Chemistry, 27.11.2019 04:31, nguyenhoangthienkim0
Acetylene gas, c2h2, can be produced by the reaction of calcium carbide and water. cac2(s) + 2h2o(l) --> c2h2(g) + ca(oh)2(aq) how many liters of acetylene at 742 mm hg and 26c can be produced from 2.54 g cac2(s)?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:30, joejoefofana
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀ pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4. 0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3


Chemistry, 22.06.2019 15:30, elizabethprasad2
The reactions of photosynthesis occur in the of plant cell? a. mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 22.06.2019 17:30, TheViperMlg23676
What causes most sediment to wash or fall into a river
Answers: 1
Do you know the correct answer?
Acetylene gas, c2h2, can be produced by the reaction of calcium carbide and water. cac2(s) + 2h2o(l)...
Questions in other subjects:


Social Studies, 27.10.2020 20:10

Arts, 27.10.2020 20:10


Social Studies, 27.10.2020 20:10

SAT, 27.10.2020 20:10



History, 27.10.2020 20:10

Mathematics, 27.10.2020 20:10


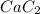 =
= 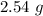
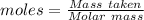
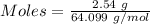
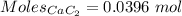
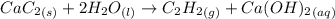
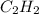
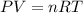
![26^oC=[26+273]K=299K](/tpl/images/0392/7554/6a452.png)