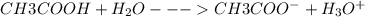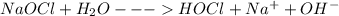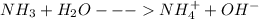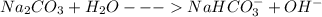Chemistry, 27.11.2019 03:31, xMABRYx1991
Write net brønsted equations that show the acidic or basic nature of the following solutions in water. for polyprotic species (vitamin c, lemon juice, and washing soda), show only one proton transfer. remember that spectator ions are not included. (use the lowest possible coefficients. omit states-of-matter in your answer.)
(a) vinegar (acetic acid, hc2h3o2)
(b) bleach (sodium hypochlorite, naocl)
(c) ammonia (nh3)
(d) vitamin c (ascorbic acid, h2c6h6o6)
(e) lemon juice (citric acid, h3c6h5o7)
(f) washing soda (sodium carbonate, na2co3)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, ashleyjaslin
Calculate the expected ph values of the buffer systems from the experiments (a, b,c, d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2


Chemistry, 22.06.2019 05:00, smartboy2296
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 07:20, rscvsdfsrysas1857
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1
Do you know the correct answer?
Write net brønsted equations that show the acidic or basic nature of the following solutions in wate...
Questions in other subjects:


Mathematics, 23.10.2019 01:00

History, 23.10.2019 01:00

English, 23.10.2019 01:00



Social Studies, 23.10.2019 01:00


Mathematics, 23.10.2019 01:00